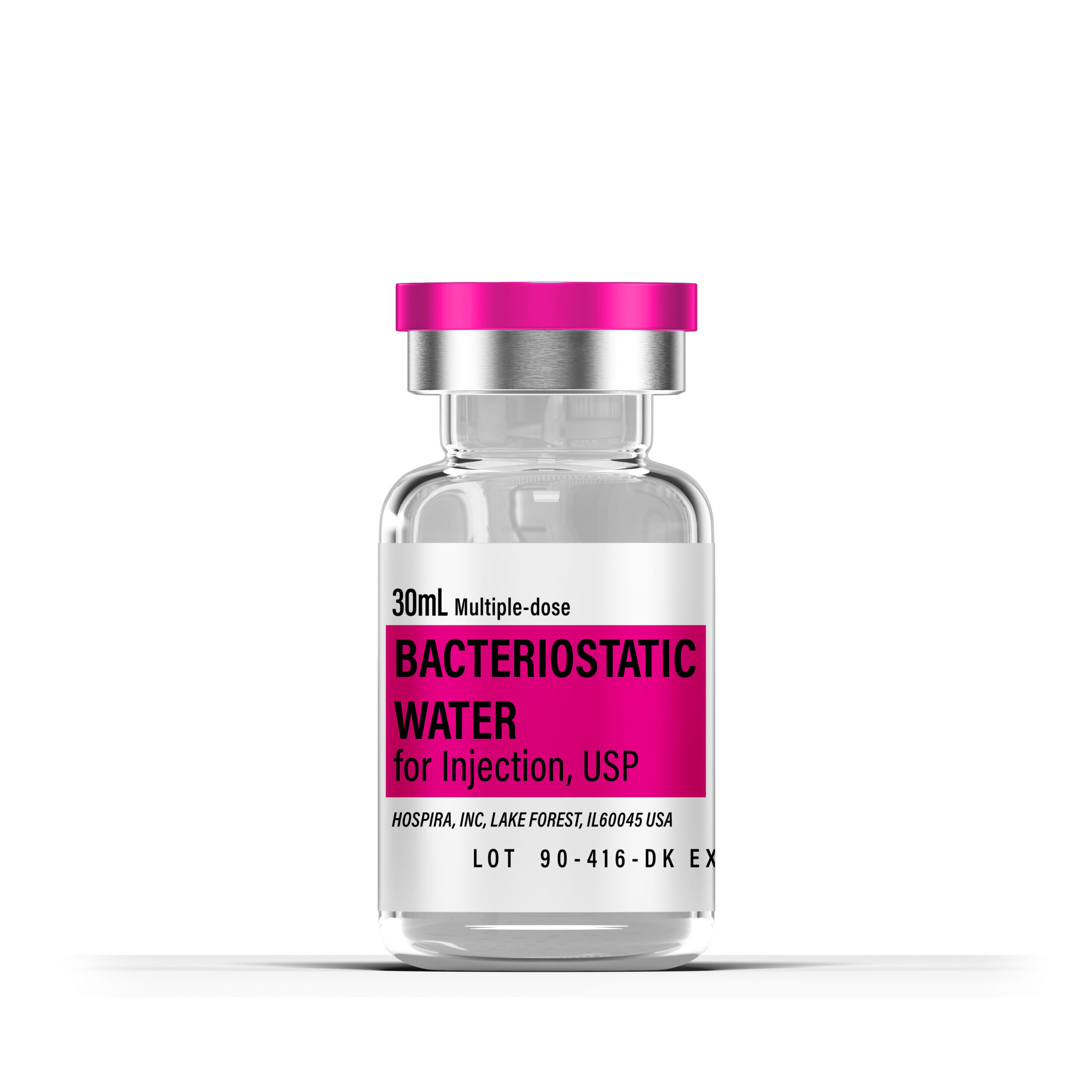
Hospira Bacteriostatic Water 30mL – Sterile Multiple-Dose Diluent Hospira Bacteriostatic Water for Injection, USP is a high-purity, sterile, nonpyrogenic preparation of water for injection containing 0.9% benzyl alcohol (9 mg/mL) as a bacteriostatic preservative, supplied in a multiple-dose vial for laboratory use in reconstituting or diluting research compounds, peptides, or other compatible reagents. This pharmaceutical-grade diluent serves as an essential tool in controlled experimental settings for maintaining sterility during repeated withdrawals, facilitating precise preparation of solutions for in vitro assays, cell culture applications, enzymatic studies, or other biochemical and pharmacological investigations requiring an aqueous vehicle.
In standard laboratory protocols, bacteriostatic water enables multiple aseptic accesses from a single vial without promoting bacterial growth due to the benzyl alcohol preservative, supporting extended experimental workflows, dose preparations, or serial dilutions while minimizing contamination risks in qualified research environments. It is commonly utilized alongside lyophilized research peptides or proteins to achieve stable, injectable-grade solutions for mechanistic studies, metabolic pathway analyses, or cellular response models, ensuring compatibility and reliability in controlled scientific setups.
The 30 mL multiple-dose presentation is well-suited for routine laboratory needs, multiple reconstitution events, larger-scale exploratory studies, or experiments requiring repeated access to diluent material. Each vial is supplied as a clear, colorless solution in a plastic or glass fliptop vial with documented sterility and purity compliant with USP standards, verified through manufacturer quality controls and third-party testing where applicable.
Specifications
- Quantity: 30 mL
- Form: Sterile solution in multiple-dose vial
- Composition: Water for Injection USP with 0.9% (9 mg/mL) benzyl alcohol
- Preservative: Benzyl alcohol 0.9%
- pH: 5.7 (range 4.5 to 7.0)
- Purity/Quality: USP grade, sterile, nonpyrogenic
- Storage: Store at 20–25°C (68–77°F); protect from light. Use aseptic technique for withdrawals. Discard after 28 days from first puncture or per protocol.
Intended Use This product is supplied strictly for in vitro and laboratory research purposes only by qualified professionals in controlled scientific environments. It is not for human or animal consumption, not for therapeutic, diagnostic, veterinary, food, cosmetic, or any non-research applications. All handling must comply with institutional biosafety and regulatory guidelines.
FDA Disclaimer The information provided on this website has not been evaluated or approved by the U.S. Food and Drug Administration (FDA). None of the statements herein, nor any products offered, are intended to diagnose, treat, cure, mitigate, or prevent any disease or medical condition. Alamo Peptides operates solely as a chemical and research reagent supplier. We are not a compounding pharmacy, do not operate under Section 503A or 503B of the Federal Food, Drug, and Cosmetic Act, are not a pharmaceutical company, and do not conduct or support clinical trials. All products are research reagents only—not for human use. By purchasing, you confirm you are a qualified researcher and assume full responsibility for lawful handling, use, and compliance.
Terms of Sale All sales are final due to the nature of research materials. Buyers must be 21+ and verify qualifications upon request. Misuse voids any implied warranties; Alamo Peptides assumes no liability for improper application. For research use only by qualified professionals. Contact support@alamopeptides.com (or via Discord/Telegram) for inquiries.



Reviews
There are no reviews yet.